
The supplement Bryan Johnson takes - and sells - didn’t survive the NIH’s longevity trial.


Hi there,
I cut a longevity supplement from my protocol this week.
Not due to cost, or even inconvenience (although the pill fatigue is real). I stopped it due to new data.
Astaxanthin was a winner in the longevity research program in 2023.
This year, the NIH's ITP longevity research program retested it — different dose, same mice — and the lifespan signal disappeared.
This is what updating protocols based on data actually looks like. It’s an important aspect of longevity medicine, which should be based on evidence which is ever-changing.
The ITP just published its 2026 results. Eight compounds tested. All failed. But the story isn't that nothing in longevity works — it's about what the failures tell us, and what's actually earned its place in the protocol.
One more thing: I'm going to show you the exact four-question framework I use to decide what stays in my protocol when data shifts. If you've ever wondered how to actually update your supplement stack rationally — not reactively — read till the end.
TL;DR
🐭 Why the ITP is different, and why most pre-clinical longevity studies aren't worth your attention
🔬 What actually works: the full shortlist of 15 compounds in 24 years, and what they have in common
🧬 AKG's second strike, two different cohorts, two nulls, and why that changes the calculus
💊 The astaxanthin paradox, passed in 2023, failed in 2026, and what the dosing gap tells us
📋 My protocol, how I actually update what I take when the data shifts
THE DEEP DIVE
The Only Pre-Clinical Longevity Screen That Actually Matters
Every week, a new study reports that some compound "extends lifespan in mice." The paper gets tweeted, the Reddit threads erupt, supplement companies update their websites — usually within 48 hours, almost always before anyone checks the methodology.
What you may not know (unless you’re a mouse scientist) is that most of those studies use inbred mice from a single strain, run at one site, by the lab that also developed the compound being tested. The results replicate maybe 30% of the time.
The Interventions Testing Program is built to fix exactly that problem.
Funded by the NIA since 2002, the ITP runs simultaneous identical experiments at three completely independent institutions: the Jackson Laboratory, the University of Michigan, and UT Health San Antonio. The mice are UM-HET3: a four-way genetically heterogeneous cross designed to approximate the genetic diversity you'd find in an actual human population. No lab knows how the other sites' mice are doing until the study ends.
Since 2002, the ITP has tested over 60 compounds. Fifteen have significantly extended lifespan.
That's a 25% hit rate, which sounds okay until you realize these compounds were pre-selected based on the strongest existing evidence. The best candidates, most compelling mechanisms, interventions most likely to work. Still, 75% fail.
What works, according to the ITP results (15 compounds in 24 years):
Compound | Effect | Notes |
|---|---|---|
Rapamycin | +9-14% both sexes | Most consistent signal; multiple cohorts |
Acarbose | +8-11% males, +5% females | Alpha-glucosidase inhibitor (diabetes drug) |
Canagliflozin | +14% males | SGLT2 inhibitor; male-specific |
17-alpha-estradiol | +19% males | Non-feminizing estrogen; castrated males strongest |
Glycine | +4-6% both sexes | Simple amino acid; inexpensive |
NDGA | +8-12% males | Plant antioxidant (not supplement-available) |
Captopril | ~5% males | ACE inhibitor |
Aspirin | +8% males | Low-dose; male-specific effect |
Astaxanthin* | +12% males | 2023 positive; 2026 retest null at lower dose |
Meclizine* | +8% males | Motion sickness drug; 2026 retest null |
Epicatechin | +10% males | Found in dark chocolate/green tea; 2025 data |
Halofuginone | +16% males | Not supplement-available; 2025 data |
The pattern is hard to ignore: mostly metabolic interventions (glucose regulation, mTOR inhibition, AMPK activation), not antioxidants. Mostly male-specific effects (ikr). The antioxidant hypothesis: the one driving most supplement marketing, has not performed well in ITP testing.
AKG: Two Strikes
Alpha-ketoglutarate has a compelling backstory.
It's a TCA cycle intermediate: a molecule your cells already use in energy metabolism and amino acid synthesis. Supplementing was theorized to "mimic caloric restriction," a mechanism with strong longevity data across species.
The original Buck Institute study showed it extended lifespan in C57BL/6 mice. The ABLE trial in humans showed improvements on epigenetic aging clocks after 7 months of use. Bryan Johnson put it in Blueprint. Peter Attia has discussed the mechanism positively. Rejuvant has been selling it at $79.95/month with explicit longevity framing.
The ITP has now tested AKG in two separate cohorts:
First cohort (2020 data): Treatment started at 18 months, deliberately matching the age protocol in the original Buck Institute positive study. Null result.
Second cohort (2022 data): Treatment started at 7 months: an earlier intervention to test whether timing was the confounder. Also null.
Two different protocols. Both null.
This doesn't mean AKG is useless across the board. The human epigenetic clock data from the ABLE trial is real. AKG may have value for muscle or bone endpoints the ITP doesn't measure, lifespan and healthspan are not the same thing, and this distinction matters.
But "extends lifespan in humans": the specific claim motivating most of its market, is not supported by the best pre-clinical evidence we have. If you're taking AKG specifically for longevity, that rationale is on shaky ground. If you're taking it for epigenetic clock modulation or other endpoints, the human data is more supportive.
Astaxanthin: The More Complicated Story
In December 2023, the ITP published results showing astaxanthin extended median male lifespan by 12%. Statistically significant, replicated across all three sites, peer-reviewed in GeroScience.
I started taking it, partially because of this study and mostly because of some other studies (below).
Then the 2026 ITP cohort retested astaxanthin. It failed.
The nuance: the 2026 test used 800 ppm in feed. The 2023 positive used 4,000 ppm. Astaxanthin has notoriously poor oral bioavailability, it's fat-soluble, the plasma concentrations are highly dose-dependent, and different formulations produce dramatically different plasma levels. At 800 ppm, you may simply not reach the concentrations that drove the original effect.
The ITP paper acknowledges this directly: "prior evidence suggesting lifespan benefits in other models or under different conditions" remains intact.
So is astaxanthin dead? No. Is the 2023 finding invalidated? Also no.
What this demonstrates is the nuanced reality of biology: we can run the gold-standard mammalian longevity trial, get a real positive, retest at a different dose, and get nothing, and now we're genuinely unsure whether the effect is dose-dependent, bioavailability-dependent, starting-age-dependent, or something else.
The drug has a real data point and a question mark next to it.
But there is real supportive human data: A 2026 systematic review of 15 human RCTs (Malcangi et al., Int J Mol Sci) found consistent reductions in IL-6, TNF-alpha, and oxidative stress markers. Anti-inflammatory and antioxidant effects in humans are real. (Effect sizes are moderate.) There's also decent data on cognitive function in older adults, cardiovascular endpoints in specific populations, and reproductive health outcomes (PCOS, endometriosis).
This is not a nothing compound. But "consistent moderate anti-inflammatory effects" and "extends lifespan" are very different claims, and I'm no longer confident the second one holds (plus I have way too many supplements). So I’m dropping this one.

THE CEO'S PROTOCOL
What I actually take, what I cut, and the framework I use when data shifts. First-person, specific — not a recommendation list.
My Protocol
I'm not going to give you a 40-item supplement list. I'm going to tell you what I'm actually optimistic about, what I cut personally and for most of my patients, and where I'm genuinely uncertain.
What I'm most optimistic about:
Rapamycin. The ITP data is the most consistent longevity signal in mammalian biology — multiple cohorts, both sexes, and the mTOR mechanism is well-understood. I'm a believer. There are risks and real contraindications, and it absolutely requires physician oversight, but for people who've done the risk-benefit math carefully, the evidence base here is stronger than almost anything else in the longevity toolkit.
Acarbose. This one is criminally underrated in most longevity conversations. It's a generic diabetes drug that blunts post-meal glucose spikes, and the ITP effect in males is strong. The sex difference in the data is real and not fully explained, but the metabolic mechanism is plausible for both sexes. Cheap, available, and the ITP has put it on the short list twice now.
Where I'm not convinced:
The SGLT2 inhibitors (gliflozins — think canagliflozin, empagliflozin) are popular in longevity medicine, and I understand why — there's a compelling signal in men in the ITP data and reasonable cardiovascular outcomes data in diabetics. But I haven't been convinced for women, particularly given the UTI and yeast infection risk profile. The risk-benefit math looks different when you're not starting from a metabolic disease baseline, and the sex-specific longevity signal in the ITP data doesn't exist for women the way it does for men. I'm watching this space, but I'm not there yet.
Side note: GLP-1 inhibitors are almost certainly lifespan-promoting, but nearly impossible at the moment to test in the ITP given its limitations (see below). But I suspect there are some similarities in the mechanism of benefits to other metabolic drugs.
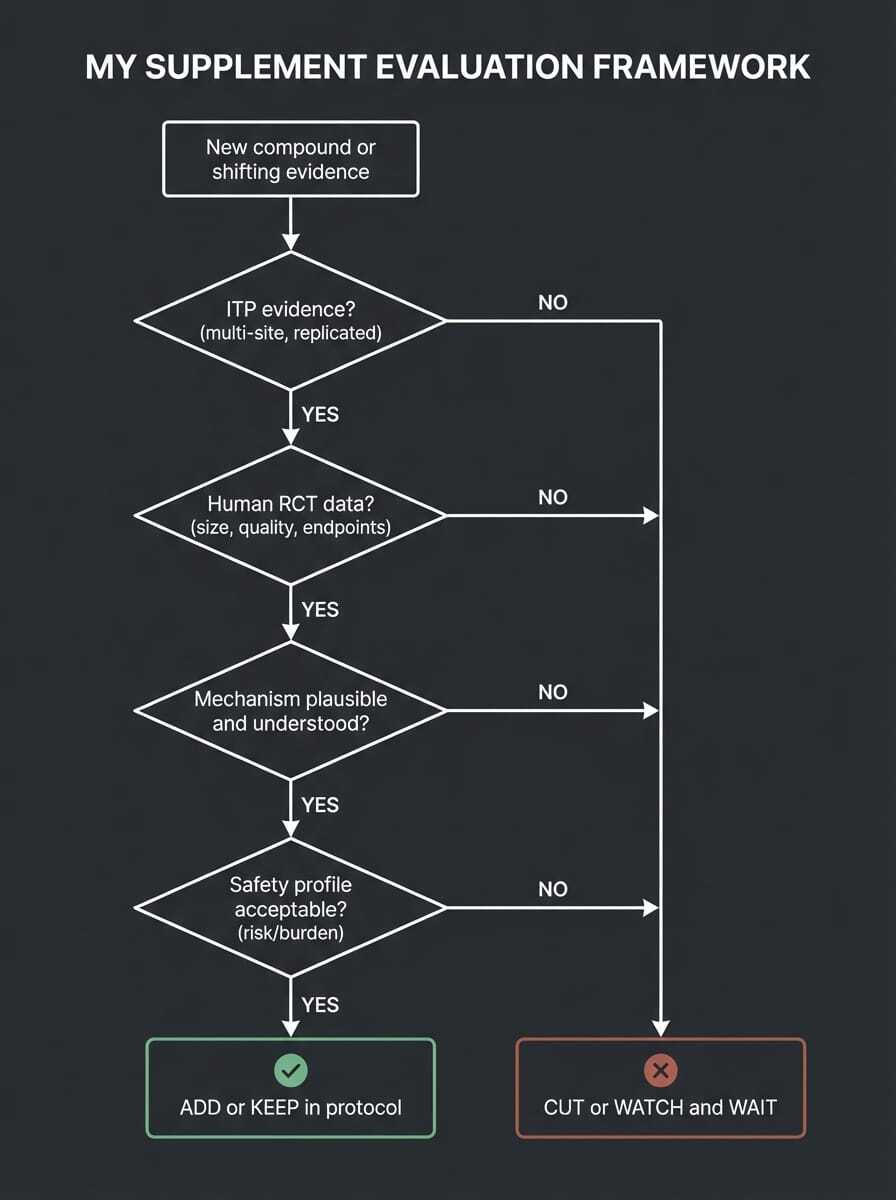
My actual framework:
When I’m considering a longevity interventions, I ask questions: What's the ITP and other preclinical evidence? What do human trials show? How strong is the mechanism? And what's the safety and burden cost of staying on it? Astaxanthin failed question one. AKG has passed question two in some limited ways but not question one. Rapamycin passes all four with appropriate caveats.
Truthfully, it is difficult to practice medicine in the longevity field, because the answers are simply not all there. I'm holding some things despite incomplete data, because mechanism is strong and safety profile is acceptable. That's a judgment call, not a formula. I'll always communicate my why to anyone who is curious.
What the ITP Can't Test
Before treating ITP nulls as verdicts, it's worth being honest about what the program is and isn't built to do.
The single biggest constraint: compounds have to be deliverable through mouse chow. They can't be injectable (no subcutaneous rapamycin analogs, for instance), they can't require medical administration, and they need to survive the food processing. This immediately excludes a huge category of compounds — anything that degrades in feed, anything that needs refrigeration, anything injectable.
The other constraint people miss: you can't test intermittent protocols. Mice eat constantly and don't fast the same way humans do. So an intervention like intermittent metformin, time-restricted rapamycin dosing, or fasting-mimicking diets can't be evaluated with any precision. Most of the interesting human longevity protocols involve how and when you take something, not just what you take. The ITP can't capture that.
And the ITP measures lifespan. Not muscle mass. Not cognitive function. Not disease burden. A compound that meaningfully preserves muscle into old age but doesn't add weeks to maximum lifespan would fail the ITP, full stop.
Use it as the best pre-clinical filter we have. Don't use it as a clinical verdict. A null in ITP mice doesn't close the human question — but it should raise your bar for confidence.
KEY TAKEAWAYS
The ITP is arguably the best pre-clinical longevity screen we have today — multi-site, genetically diverse mice, no investigator conflict.
In 24 years, 15 of 60+ compounds have worked. Rapamycin and acarbose are the most consistent. Glycine is underrated.
Astaxanthin passed in 2023 (4,000 ppm → +12% male lifespan). Failed in 2026 at 800 ppm. The dose gap matters — this isn't a verdict on the compound, it's a precision problem.
AKG has failed the ITP in two separate cohorts. If you're taking it for lifespan extension, the rationale is on shaky ground. Human epigenetic clock data is more interesting but more limited.
The pattern across ITP positives: metabolic pathway modulation, not so much antioxidants. Males generally benefit more than females (alas!).
QUICK HITS
🧬 How fast your epigenetic clock changes predicts death better than how old it reads. A new Nature Aging study tracked people longitudinally and found that the rate of change in epigenetic clocks over time outperforms a single clock snapshot for predicting mortality. Implication: a one-time biological age test tells you less than repeat testing over months or years. This is the argument for tracking, not just testing.
💊 First senolytic drug clears Phase 1 in humans. Rubedo Life Sciences just released preliminary Phase 1 data on RLS-1496, a first-in-class GPX4 modulator designed to selectively kill senescent cells, in psoriasis, eczema, and skin aging patients.
💉 Semaglutide failed two Phase 3 Alzheimer's trials. Two large trials of oral semaglutide in mild Alzheimer's disease showed no slowing of clinical progression versus placebo over two years. The GLP-1 hype as a neuroprotective agent took a significant hit. The metabolic benefits are real; the dementia prevention story is not yet.
🐭 70% lifespan extension in elderly mice with a drug combo. A December 2025 paper reported a simple drug combination (OT+A5i or oxytocin and an Alk5 inhibitor) boosted remaining lifespan by 70% in already-old male mice.
🧬 New epigenetic clock spots cancer-specific methylation drift, not just aging. A Clinical Epigenetics paper published this week developed a clock that simultaneously measures biological aging and cancer-associated methylation changes, which look distinct from normal aging patterns. Getting closer to blood-based early cancer detection via methylation.
😔 Depression accelerates biological aging. Anxiety doesn't. A Molecular Psychiatry paper showed depression independently accelerates epigenetic aging across multiple clocks, while generalized anxiety did not produce the same signal. Proposed mechanism: chronic cortisol elevation and HPA axis dysregulation driving CpG methylation at aging-associated sites. Treating depression is a longevity intervention.
🩸 ApoB just got a JAMA cost-effectiveness endorsement. A JAMA analysis co-authored by ApoB pioneer Allan Sniderman found ApoB-guided cardiovascular prevention is more cost-effective than LDL-C or Non-HDL-C targets at a population level. If you're not tracking ApoB, you now have a health economist's argument for it alongside the clinical one.
🏋️ Cardiorespiratory fitness is an epigenetic disease-risk editor. A new Aging Cell paper showed higher VO2 max is inversely associated with DNA methylation-based risk scores across the aging disease spectrum simultaneously, not just heart disease. One fitness metric, system-wide epigenetic benefit.
THE BIGGER PICTURE
The ITP 2026 results are easy to misread as a failure of longevity science. They're the opposite.
When a compound passes in one dose and fails at another, we've learned that the therapeutic window is narrow, bioavailability is likely the confound, and we need better pharmacokinetic characterization before making clinical recommendations. That's science working exactly as designed.
The harder lesson is what this means for how we communicate about supplements. The standard in longevity medicine should be strong preclinical evidence, human trial data, mechanism plausibility, safety profile, and a clear-eyed calculation of supplement burden versus demonstrated benefit. Not influencer endorsements. Not single-lab mouse studies that haven't been replicated. Not cellular mechanisms that sound plausible but haven't cleared a rigorous screen.
We need more ITP-style programs. More multi-site replication. More transparency when findings don't hold.
Longevity needs to move from hope, to science.
FROM THE LONGEVITY SHOW ⭐

Rational Biohacking: A Framework for Scientific Longevity and Wellness
The framework I use to decide what stays in my protocol — and what gets cut — is something I've built over years of clinical practice and digging into the evidence. I broke it down in full in this episode. If the ITP data made you want a cleaner system for evaluating longevity claims, this is the place to start. The four questions I described above? This is where they came from.
Also, as a bonus because I’m so excited about it, I just dropped Part 2 of our Lipid Series on YouTube. I spent a long time researching and planning this one, so excited to hear your thoughts. Check it out here!
Keep learning,
Hillary Lin, MD
Co-Founder & CEO, CareCore
Was this useful? Forward The Longevity Letter to someone who would benefit.
Want to turn your wellness brand into a full-service health destination? Learn about CareCore's platform or Get Started Here
Where to find me
👱🏻♀️👩🏻🦰👩🏻👧🏽👧🏾 Livelong Women's Health Summit — April 17-18, 2026, SF, CA. Delighted to join 50 other thought leaders in speaking on women's longevity! (Code HILLARY50 for $50 off)
🎤
The Aging Code Summit, May 26-27, Mosesian Center for the Arts, Greater Boston. I'll be on the clinician panel discussing how aging research translates into real interventions.
Dry Eye Society of the Americas, July 10-11, 2026, NYC. Dry eyes are a growing problem and symptom of deeper issues tied to longevity. For clinicians - join us.
Support us with your longevity purchases
🐟 Seatopia — sustainable, sashimi-grade seafood checked for microplastics and mercury (Code HILLARYLINMD for $20 off)
⏱️ Timeline — patented Urolithin A for scientifically proven mitochondrial support via mitophagy (Code CARECORE for 10% off)
🍺 ZBiotics — pro-longevity probiotics for when you do have a little fun (Code CARECORE for 10% off)
🧲 CoreViva — whole-body MRI screening for early disease detection (Code CARECORE for $200 off)
🧠 NeuroAgeTx — the most comprehensive brain aging package at up to 61% off for Longevity Letter readers
🧬 GlycanAge — measures your immune system's biological age and recommends how to fix it
Advanced diagnostics
Want to know your biological age? Check out our testing page for epigenetic age testing with physician consultation, including TruDiagnostic TruAge and SystemAge by Generation Lab.
Have a question or topic you want covered? Reply to this email.
Was this forwarded to you? Subscribe here to get The Longevity Letter every week.
Next step
Get the physician’s note, not the wellness-script version.
The Longevity Letter turns the same clinical filter into a weekly read: what is worth measuring, what is worth changing, and what is just noise.